The comments on a previous post indicated some understandable confusion on the part of some commenters as to the relationship (or lack thereof!) between various measures of “stability” in ecology. The term “stability” is infamous for meaning different things to different people, so that entire areas of the literature, particularly on the links between “diversity” and “stability”, are rife with confusion. Previous attempts to clear up the confusion (e.g., Pimm 1984) seem not to have had much long-term effect, so odds are that no blog post of mine is going to help much. But long odds of doing any good have never stopped me from posting before. 😉
Below is a list of every “stability” concept that I could think of off the top of my head last night, with a brief definition, a useful reference or two (sometimes to the paper defining the concept, sometimes just to an arbitrarily-chosen paper illustrating or applying the concept), and perhaps a few brief interpretive remarks.*
The single most important message you should take away from this list is that these different kinds of stability are different, and in many cases have little or nothing to do with one another. Even when they are related, it’s often in complex, non-intuitive ways. For instance, to pick just one possible example of many, the same conditions that synchronize the fluctuations of the abundances of different species (thereby increasing the temporal variability or “instability” of their total biomass) also can cause their fluctuations to decrease in amplitude and be bounded further from zero, both of which are “stabilizing” (Vasseur and Fox 2007). So the correct answer to the question “Do synchronizing factors decrease or increase stability?” is “What exactly do you mean by ‘stability’?”
I’ve grouped the different stability concepts into three rough categories, although the last category is a catch-all for stability concepts that don’t really fit with anything else.
I have not made any comments on the underlying drivers or causes of any of these stability measures. This post is just about clarifying concepts. Oikos is not paying me nearly enough** to try to write a post summarizing everything that’s ever been written on the drivers of all these different things!
You can talk about the stability of anything that can change over time, but for concreteness I’ll present definitions couched in terms of community ecology.
Cranky note to readers who are not mathematically inclined: This list is only an entry point into the literature. It is no substitute for reading and understanding the literature. In most cases this will oblige you to learn some math. I know that’s probably not what you want to hear, but that’s the way it is. Many important concepts of “stability” are mathematical. It is not possible for me, or anyone, to properly explain these concepts using only words. Math is more precise than words, so translating math into words always represents a loss of information and an increase in ambiguity. Words have multiple meanings, and “stability” is no exception. If you don’t know what meaning is being used in a particular context, there are going to be tears before bedtime. And while some theoreticians sometimes could do a better job of explaining themselves, there’s a limit to how much it’s possible for them to walk you through this stuff. Theoretical papers in the primary literature necessarily assume some mathematical expertise on the part of the reader, just like a paper reporting field experiments necessarily assumes some expertise on the part of the reader with things like experimental design and statistics. So if you want to understand this stuff well enough to work on it or teach it well, you’re going to need to put the effort in to learn some math.
So if you’re the kind of person who reads theoretical papers by skipping the equations and just reading the words, because you just want to “get the gist”, I’m sorry, but it’s pretty much inevitable that you’re going to end up confused about stability. When it comes to “stability” (and many other important ecological concepts), there either is no ‘gist’ to get, or the only way to get it is to first get the technical details. You’d be appalled, and rightly so, if some theoretician said to you, “Plants and algae are basically the same, they’re all green, the differences are just unimportant technical details that only plant ecologists need to worry about”. Which is exactly how big a mistake you’re making if you think that “All these different kinds of stability are basically the same, they’re all related, the differences are just unimportant technical details that only theoreticians need to worry about”. Yes, I know Charles Elton (1958) conflated several different stability concepts in his very influential book on invasion ecology. But he had the very good excuse of writing many decades ago. You don’t have that excuse, and shouldn’t follow his example.
And if you say, “But I don’t know enough math to really understand the details of different stability concepts,” then your choices are to learn some math, or work on something else. Just like how, if you don’t know enough about plants to really understand the plant ecology literature, your choices are to either learn more about plants, or work on something else.
And you know what? It can actually be fun to learn this stuff! Seriously. It’s like anything that takes a bit of effort to appreciate—once you get into it a little, it’s pretty cool.
Note to mathematically-inclined readers: Yes, many of my definitions are very imprecise. Please don’t hassle me in the comments. I am aware of more precise definitions. I chose to write the post in this way because it’s only an entry point into, and rough road map of, the literature, aimed at non-mathematicians. My imprecision will do no harm. No one is going to rely solely on my definitions. I’m only trying to be precise enough to give non-mathematical readers a sense of just how many different stability concepts there are, and just how different they are from one another.
Stability concepts related to equilibria and other attractors
These are the stability concepts that tend to get used most often by theoreticians.
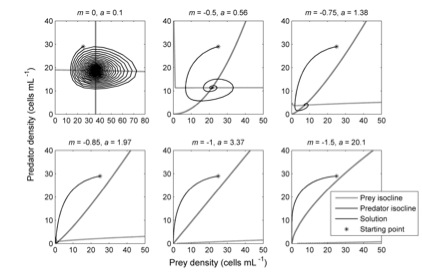
Feasibility of equilibrium, and interior vs. boundary equilibria. Is there a set of non-zero densities at which all species have zero population growth rates? If so, the system has a feasible “interior” equilibrium. An equilibrium at which one or more species have zero density is known as a “boundary” equilibrium. An equilibrium at which one or more species have negative densities is “infeasible”, because negative densities are physically impossible.
Stability of equilibrium. Will species’ densities approach a given equilibrium over time if they’re not currently at that equilibrium (for instance, because they’ve just been perturbed away from their equilibrium values)? Equilibria that are stable in this sense are sometimes called “resilient”. The opposite is an unstable equilibrum, an equilibrium state that the community tends to move away from, rather than towards. A neutrally stable equilibrium is one the community tends to move neither towards nor away from.
(Asymptotic) rate of return to equilibrium. How fast does the community return towards, or move away from, an equilibrium state? Typically, theoreticians ask about the asymptotic rate of return, meaning how fast the system returns once it is already sufficiently close to equilibrium (systems that are sufficiently close to a stable equilibrium approach that equilibrium at a constant rate). This is an asymptotic rate because, in general, if a community is far from a stable equilibrium its rate of return to that equilibrium can fluctuate greatly over time, and can even be temporarily negative (see “reactivity”). But eventually (“asymptotically”) the system gets close enough to equilibrium to approach it at a constant rate.
Local vs. global stability of equilibrium. A community that returns to an equilibrium following a sufficiently-small perturbation (i.e. a perturbation that only changes species’ densities by a sufficiently small amount from their equilibrium values) is said to be locally stable. A community that returns to equilibrium following any possible perturbation that doesn’t actually eliminate a species is said to be globally stable.
Domain of attraction. How far can you can perturb species’ densities away from equilibrium and still have them return to that equilibrium? By definition, a globally stable community has the largest possible domain of attraction.
Alternate stable states. A community with multiple, locally-stable equilibria has alternate stable states. Which equilibrium the community approaches in the long run depends on its initial state. For instance, a community that starts out close to one equilibrium might approach that one rather than another, more distant equilibrium.
Attractor. Any state or sequence of states which a system tends to approach if it’s not currently in that state. Stable equilibria are one kind of attractor. Others include stable limit cycles and chaotic attractors. Most of the stability properties of equilibria are also properties of other kinds of attractors too. For instance, attractors can be local or global attractors, a community can have alternate local attractors, etc. Also, some kinds of attractors are sometimes considered as more “stable” than other kinds (e.g., equilibria>limit cycles>chaos).
Permanence. A community is permanent if the community tends to move away from boundary equilibria. Basically, this means that, if one or more species are currently rare, they tend to increase rather than decline to extinction. Permanence implies the existence of some sort of interior attractor.
Reactivity. A system is reactive if, on being perturbed away from a stable equilibrium, it initially moves even further from that equilibrium before eventually returning.
Probability of local asymptotic stability (or feasibility, or permanence, or etc.). Given a specified model of system dynamics, with parameters randomly chosen from specified distributions, what is the probability that the resulting system will have a locally-stable interior equilibrium, or have a feasible interior equilibrium, or be permanent, or be reactive, or etc.?
Sign stability. An equilibrium which is guaranteed to be stable, just due to the signs of the interactions among the species (e.g., predators have a negative effect on prey growth rate, while prey have a positive effect on predator growth rate), no matter what the absolute magnitudes of those interactions.
References: May 1973 (stability of equilibrium, sign stability, probability of stability), Goh and Jennings 1976 (feasibility), Law and Morton 1996 (permanence), Neubert and Caswell 1997 (reactivity), Case 2000 (entry-level textbook covering various stability concepts related to equilibria and attractors)
Stability concepts related to variability
These are the stability concepts that, at least lately, are most often used in empirical studies, although there is a fair bit of theoretical work as well.
Temporal variability of a single variable. How much the abundance of a given species, or some other variable of interest, varies over time. Can be measured by various statistics, which themselves differ from one another (e.g., variance, coefficient of variation). Some authors use “constancy” to refer to the property of having low temporal variability.
Temporal variability of the sum of a set of variables. How much the total abundance or biomass of a set of species, or the sum total of some other set of variables, varies over time. The answer depends both on how the individual variables (the summands) vary, and on how they covary. All else being equal, negative covariation among the summands reduces the variability of their sum. Can be measured by various statistics, which themselves differ from one another (e.g., variance, coefficient of variation).
Range or amplitude of variation. The range of values over which the variable of interest fluctuates (maximum minus minimum). For variables that oscillate (cycle) in a periodic fashion, this is known as the amplitude of the oscillation.
Stationarity. A stationary variable is one that fluctuates over time, but with unchanging mean, variance, and other statistical moments. So for instance, a species that’s gradually declining towards extinction (i.e. mean abundance is declining) is not stationary.
References: Ives and Hughes 2002, Loreau and de Mazancourt 2008
Other stability concepts
Resistance. A system is resistant if it’s difficult to perturb it away from its current state. For instance, a small fire that kills grassland plants (thereby perturbing their abundances) might have no effect on tree abundances, indicating that trees are more resistant than grass to small fires.
Species deletion stability. If you remove a species from a community without changing anything else, does that lead, directly or indirectly, to any other species going extinct? If not, the system is species deletion stable. If so, then you have “secondary extinctions”, which could themselves lead to further extinctions (an “extinction cascade”).
Network topology stability. If you have a network of dependencies (e.g., predators are linked to, and depend on, their prey, and plants and pollinators are linked to, and depend on, each other), and you start removing species and/or links from that network, how many and/or which links do you have to remove in order to remove all the links on which a given species, or any species, depends? I made up the name for this, there doesn’t seem to be an agreed term in the literature.
Invasion resistance. If you add a new species to the community, at initially-low abundance, can it increase and establish itself? If not, the community is invasion resistant. Actually, this is related to local stability of boundary equilibria, so maybe it doesn’t belong in this subsection…Note that, in the theoretical literature, an “invader” is just an initially-rare species. It’s not, or not necessarily, “non-native” or “exotic”. In defining “invader” in the way that they do, theoreticians are focusing on the ecological determinants of invasion success. After all, species move around and establish new populations in new locations all the time, and they always have. Whether or not “non-native” (or “exotic” or “weedy” or etc.) species tend to have the properties that allow them to invade is a separate question; the mere fact that a species is “non-native” or “exotic” does not in and of itself affect its ability to invade.
Boundedness away from zero. How closely does the abundance of a given species, or some other variable that can only take on non-negative values, approach zero? The less closely it approaches zero, the more “stable” it is reckoned to be, on the basis that random events are more likely to cause the variable to actually go to zero if it closely approaches zero on its own.
Persistence time. How long does a species, or the community, persist before that species, or one or more of the species in the community, goes extinct?
References: Pimm 1980 (species deletion stability; an Oikos classic!), Case 1991 (invasion resistance), McCann et al. 1998 (boundedness away from zero)
*I wish I could just direct readers to Wikipedia for this, but the Wikipedia page on “ecological stability” is poor. The section on local vs. global stability says “Local stability indicates that a system is stable over small short-lived disturbances, while global stability indicates a system highly resistant to change in species composition and/or food web dynamics.” Huh? A locally stable system is one that’s…stable? Local stability has something to do with small disturbances, whereas global stability has to do with large disturbances resistance to change? Other sections are just as bad. I often find Wikipedia useful, but this isn’t one of those times. It’s not that the page is brief, it’s that what’s there is confusing or wrong.
**Indeed, they’re not paying me anything.














































































































![Male of the harvestmen Zygopachylus albomarginis (with yellow ink marks) inside his mud nest, while a female approaches from the outside [Credit: Gustavo S. Requena]](https://oikosjournal.wordpress.com/wp-content/uploads/2014/09/fig1.jpg?w=300&h=225)
![At the left, we can see a male inside his nest on a fallen trunk without fungus infestation. At the right, the trunk is covered by fungus fruiting bodies, except inside the nest. Nest-cleaning behavior maintains hygienic conditions inside the nest at the same time it provides food to the male, which feed upon the fungus hyphae. [Credit: Gustavo S. Requena]](https://oikosjournal.wordpress.com/wp-content/uploads/2014/09/fig2.jpg?w=500)
![Male resting inside his nest, which contains several black eggs (indicating advanced embryonic development) [Credit: Gustavo S. Requena]](https://oikosjournal.wordpress.com/wp-content/uploads/2014/09/fig3.jpg?w=300&h=225)